Innovation
Innovation
What is WaveSense™ Technology?
WaveSense™ is a suite of patented technologies that uniquely applies Dynamic Electrochemistry in blood glucose measurement to detect and correct for common sources of error, providing accurate results. WaveSense Technology is included in all AgaMatrix Meters.
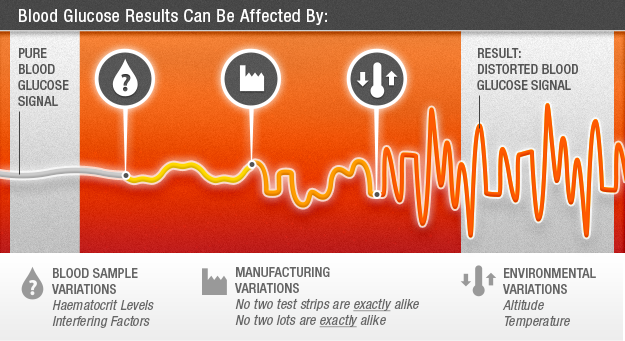
What Can Cause Distortions in the Blood Glucose Signal?
The blood glucose signal can be affected by variations in blood samples, environmental conditions, and manufacturing processes. These distortions can lead to inaccurate blood glucose measurement.
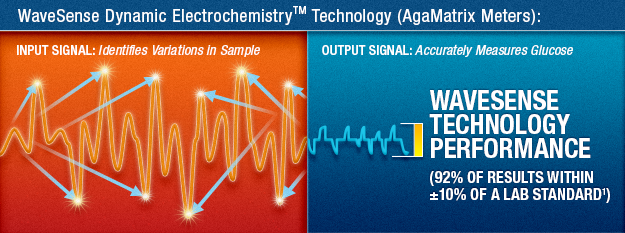
How WaveSense™ Dynamic Electrochemistry® Works
WaveSense Dynamic Electrochemistry is able to detect and correct for common sources of distortions, providing accurate blood glucose measurements.
First, complex electrical signals are sent into the test strip to identify variations in the sample. Then advanced digital signal processing algorithms are used to correct for many errors caused by variations in blood samples and environmental conditions.
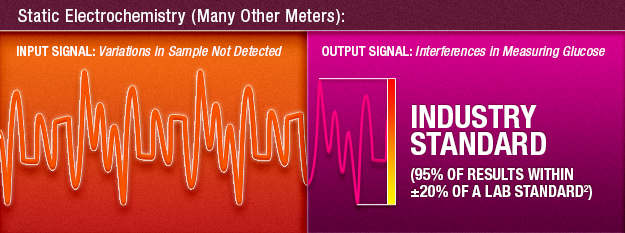
Static Electrochemistry (Many Other Meters)
Many other meters use Static Electrochemistry, which sends a simple constant signal into the test strip–typically ignoring distorting factors.
Why Accuracy Matters
Diabetes management starts with a blood glucose reading–what if it was off by 20%? Since diabetes treatment decisions are based on a blood glucose reading, accuracy can affect everything from how much insulin is taken to diet choices and even patient safety.
The chart below shows how varied a blood glucose reading can be at ±20% or ±10% compared to the actual blood glucose (laboratory result).
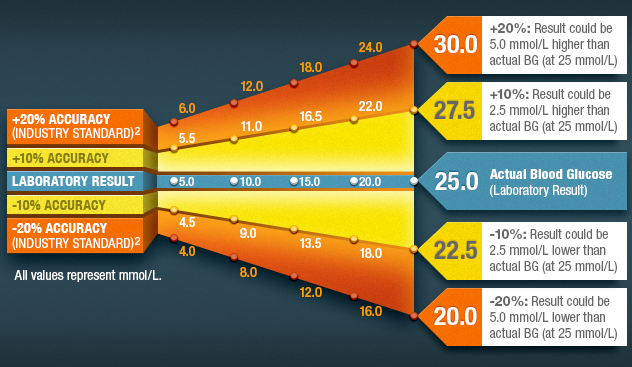
The current industry standard (EN ISO 15197:2015) accuracy for blood glucose meters allows for ±15% variation from a laboratory standard.
Clinical studies show that the AgaMatrix WaveSense JAZZ Meter provides results within ±15% from a laboratory standard 99% of the time, meeting the EN ISO 15197:2015 criteria.*
*Based on an ISO 15197:2013 system accuracy study; YSI reference.
1. Based on an ISO 15197:2003 system accuracy study; YSI reference.
2. ISO 15197:2003 In vitro diagnostic test systems —Requirements for blood-glucose monitoring systems for self-testing in managing diabetes mellitus
3. EN ISO 15197:2015 In vitro diagnostic test systems —Requirements for blood-glucose monitoring systems for self-testing in managing diabetes mellitus
Apple and the Apple logo are trademarks of Apple Inc., registered in the U.S. and other countries. App Store is a service mark of Apple Inc.
All trademarks and/or copyrights are properties of their respective owners.
